|
|||||||||||
|
|
|||||||||||
|
Feature Articles: Creating Novel Functional Materials Vol. 17, No. 10, pp. 7–11, Oct. 2019. https://doi.org/10.53829/ntr201910fa2 Creation of Novel Material Sr3OsO6 with the Highest Ferromagnetic Transition Temperature among InsulatorsAbstractWe have synthesized a novel material, Sr3OsO6 (Sr: strontium, Os: osmium, O: oxygen) using a unique oxide thin-film growth technique that has been developed over many years at NTT Basic Research Laboratories. The Curie temperature (TC) value of this material, estimated from the magnetic measurements, is above 780°C, surpassing the TC record among insulators for the first time in 88 years by more than 100°C. As Sr3OsO6 has been synthesized in a single-crystalline thin film form, this brand-new material is expected to be readily implemented in high-performance magnetic device applications such as magnetoresistive random access memories and magnetic sensors that work above room temperature. Keywords: molecular beam epitaxy, magnetic oxide, spin-orbit coupling 1. IntroductionFerromagnetism is a magnetic state in a material that gives it the properties of a magnet. In the ferromagnetic state, the net magnetic moment is large since the magnetic moments of the constituent atoms are aligned (Fig. 1). A ferromagnetic insulator is a magnet in which electric current cannot flow due to its high resistivity. Ferromagnetic insulators include maghemite, the first magnet that humans discovered and used as a compass. Today, ferromagnetic insulators are widely used as permanent magnets and in the microwave devices incorporated into, for instance, smartphones, cars, and computers—and such technology could not have been developed without ferromagnetic insulators. Spintronic devices, in which both the electrical and magnetic properties of electrons are utilized simultaneously, are now being extensively investigated to achieve high-speed devices with low power consumption. Ferromagnetic insulators will also serve as essential constituents that make such spintronic devices viable.
In conjunction with trends in computerization, there has been a steadily growing demand for practical devices with higher performance. In terms of temperature, stable operation even above 200°C is required. However, the record Curie temperature (TC), which is the crucial factor determining the temperature range in which any ferri/ferromagnetic system remains stable, has stood in insulators ever since ferrite magnets*1 were first developed over eight decades ago in the 1930s. Therefore, researchers have sought to develop the next generation of ferromagnetic insulators with high TC values as well as to establish guiding principles to search for such materials.
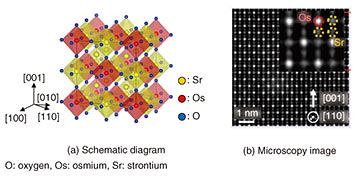
2. Preparation of high-quality Sr3OsO6 thin filmsSolids in which atoms are periodically and orderly arranged and thus form lattices are called crystals. Crystals that have only a single atomic arrangement over an entire volume are known as single crystals. Samples whose thicknesses range from one atomic layer to about several tens of micrometers (1 μm = 1 × 10–6 m) are called thin films. Single-crystalline thin films are synthesized on single-crystalline substrates. For microfabrication of high-performance devices, it is necessary to prepare samples in the form of single-crystalline thin films with submicrometer thicknesses. In this study, single-crystalline Sr3OsO6 thin films with a thickness of 300 nm (1 nm = 1 × 10–9 m) were synthesized on single-crystalline SrTiO3 (strontium titanate) substrates [1]. A schematic diagram of the double perovskite structure of Sr3OsO6 is shown in Fig. 2(a). The yellow, red, and blue spheres respectively indicate Sr (strontium), Os (osmium), and O (oxygen) atoms. Sr3OsO6 is a novel material synthesized in this study for the first time. In crystals, atoms are regularly ordered and form lattices. There are many kinds of atomic arrangements (crystal structures), and representative ones have specific names. One such arrangement is known as double perovskite, in which the lattice is twice as large as the perovskite structure. Many complex oxides are known to have the perovskite structure. Iodides and chlorides that have the perovskite structure have also been extensively studied recently to develop the next generation of solar cells [2]. To grow high-quality Sr3OsO6 thin films, precise control of the flux rate of each constituent cation (Sr, Os) is mandatory. Generally, controlling the flux of Os is a challenge because of its high melting point (3033°C). Nevertheless, we have succeeded in precisely controlling both the Sr and Os flux rates. We accomplished this by monitoring the flux rates with an atomic emission spectrometer and feeding them back to the evaporation source power supplies in real time, which enabled the synthesis of Sr3OsO6 thin films with the Sr and Os atoms arranged in a highly ordered structure. An atomic resolution microscopy (scanning transmission electron microscopy) image of an Sr3OsO6 film viewed along the [110] direction is shown in Fig. 2(b). We can clearly see the atomic ordering depicted in Fig. 2(a).
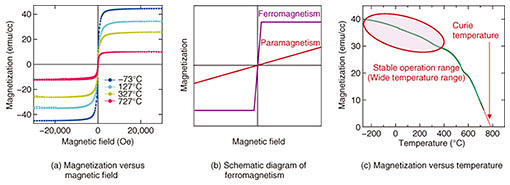
3. Ferromagnetism above 780°C in Sr3OsO6As described above, we have synthesized the novel material Sr3OsO6 having the highest TC among insulators by using a unique oxide thin-film growth technique that we have developed over many years. First, we measured the electrical and optical properties of the Sr3OsO6 thin films. The resistivity at room temperature was 75 Ω·cm, which is about 109 times as large as typical metals such as Au (gold) and Cu (copper). Also, resistivity increased exponentially as the temperature decreased. Furthermore, the optical band gap of Sr3OsO6 was found to be about 2.65 eV. All these results indicate that Sr3OsO6 is an insulator. Next, we examined magnetic properties. The magnetization versus applied magnetic field curves of an Sr3OsO6 film is shown in Fig. 3(a). It shows ferromagnetic behavior (Fig. 3(b)) with a finite magnetization even at the high temperature of 727°C. The magnetization versus temperature curve of an Sr3OsO6 film is shown in Fig. 3(c). The applied magnetic field was 2000 Oe. The gradual change in the magnetization up to about 400°C is suitable for high-performance magnetic devices that can be stably operated at high temperatures (above room temperature). The TC value, at which ferromagnetism disappears, is above 780°C, surpassing the TC record among insulators for the first time in 88 years by more than 100°C.
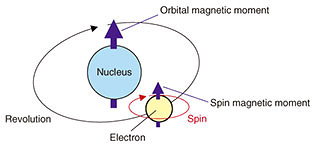
In addition to the experiments, density functional theory*2 calculations were carried out by the Tsuneyuki Research Group at the University of Tokyo. These calculations revealed that the ferromagnetic insulating state of Sr3OsO6 originates from the large spin-orbit coupling of the 5d element Os. The spin-orbit coupling consists of interactions between the spin magnetic moment coming from the axial rotation of electrons and the orbital magnetic moment coming from the revolution of the charged particles (electrons) around the nucleus (Fig. 4). The elements in the lower rows of the periodic table are heavier than those in the higher rows, and they have a larger spin-orbit coupling. Thus, the spin-orbit coupling in Os is larger than in Fe (iron) and Co (cobalt), which are used in typical magnets. This insight into the mechanism of the emergent high-temperature ferromagnetism will open a new avenue for developing functional materials in which elements having large spin-orbit coupling play a role.
This novel material Sr3OsO6 has been synthesized in the form of single-crystalline thin films, which have high compatibility with device fabrication processes. This is in marked contrast to typical new oxides often synthesized in a powder or sintered polycrystalline form. Thus, Sr3OsO6 is expected to be readily implemented in high-performance magnetic device applications such as magnetoresistive random access memories (MRAMs) and magnetic sensors that work above room temperature.
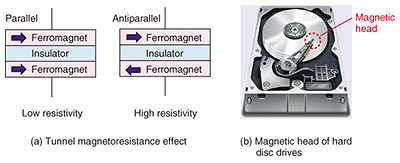
4. Future outlookIn our quest to better understand the fundamentals of ferromagnetism, we will further investigate the electronic structures of Sr3OsO6 using advanced spectroscopy techniques provided by synchrotron radiation facilities.*3 As part of efforts to develop high-performance magnetic devices that can be operated at high temperatures, we are working on fabricating some test devices using Sr3OsO6 to examine the tunnel magnetoresistance effect. This effect occurs when the tunnel resistance of an insulator sandwiched between two ferromagnets changes depending on the magnetic configuration of the ferromagnets (parallel or antiparallel) (Fig. 5(a)). The tunnel magnetoresistance effect has been widely utilized in commercial devices such as hard disc drives (Fig. 5(b)), MRAMs, and magnetic sensors. Therefore, demonstration of the tunnel magnetoresistance effect in Sr3OsO6 will lead to progress in developing high-performance magnetic devices that work at high temperatures.
References
|
|||||||||||