|
|||||||||||||||||
|
|
|||||||||||||||||
|
Feature Articles: Research and Development toward Sustainable Infrastructure Accelerated Aging Testing of Plastic Materials to Extend Service Life of Telecommunications InfrastructureAbstractPlastic materials that are used outdoors deteriorate due to the combined effects of light, heat, water, and stress. Therefore, evaluation techniques that enable the identification and use of materials with excellent weather resistance are essential for extending the lifespan of infrastructure facilities. In this article, we introduce our efforts to establish accelerated aging tests for appropriately evaluating weather resistance, using polypropylene as an example. Keywords: plastic materials, weather resistance, accelerated aging tests 1. Plastic materials and their degradation in telecommunications infrastructurePlastic materials are key components of telecommunications infrastructure, including cable sheathing, branch cable covers, closure housings, and bundling materials. While plastic materials have advantages such as being lightweight and having excellent formability, corrosion resistance, and electrical insulation, they are more susceptible to deterioration in appearance and properties due to changes in their chemical structure caused by light and heat than inorganic materials such as metals, ceramics, and glass [1, 2]. In the field, the rate of material degradation is strongly affected by the environmental conditions in which they are used due to the combined effects of factors such as ultraviolet (UV) light, temperature, rainfall/condensation, wind, and pollutants [3]. Therefore, to extend the life of telecommunications infrastructure by developing materials that have the required characteristics that can effectively counter degradation, appropriate performance-evaluation tests must be conducted on the basis of the usage environment. Therefore, we are conducting research on plastic materials intended for exposure to outdoor environments to establish a unique accelerated degradation test for accurately evaluating material properties by reproducing the degradation phenomena that occur in real environments while accelerating degradation. In this article, we introduce our work on accelerated aging testing using polypropylene, a material widely used in telecommunications infrastructure, as a case study. 2. Significant deterioration factorsPlastic degradation involves molecular-level chemical reactions such as oxidation, decomposition, and crosslinking, resulting in various changes in physical properties and characteristics such as embrittlement, whitening, cracking, and reduced strength. As mentioned above, in outdoor environments, degradation is caused by the synergistic effects of light, heat, and water, making it difficult to isolate individual phenomena. We provide an overview of the most significant degradation factors as follows. The first major degradation factor is sunlight (UV rays). Photodegradation occurs when the UV rays in sunlight break chemical bonds in the plastic, generating radicals*1, which, in the presence of oxygen, lead to a chain reaction of photooxidation. Ideally, many general-purpose plastics, such as polypropylene, absorb very little sunlight. However, trace amounts of impurities, catalyst residues*2, or trace amounts of carbonyl groups can absorb light energy, causing photodegradation [1]. Compared with the molecular bond energy of polymers, light energy in the UV range can be sufficient to break bonds. Therefore, in outdoor environments where there is a large amount of UV light, oxygen-mediated photooxidation reactions are likely to occur, leading to surface discoloration, chalking*3, embrittlement, and cracking. Next is heat. Heat increases the rate of chemical reactions, accelerating light-induced oxidation reactions. It has been reported that the simultaneous action of light and heat can synergistically accelerate deterioration [1, 2]. When exposed to high temperatures in the atmosphere for long periods, the heat and oxygen act to promote autoxidation reactions, which proceed in a chain reaction while generating oxygen-containing functional groups such as hydroperoxide, carbonyl, and hydroxyl groups. These reactions progress more easily at higher temperatures, and heat diffuses from the surface to the interior of the material, degrading even thick components [1]. Water is also an important factor. In environments where materials come into contact with moisture, such as rain, frost, condensation, or immersion, additives near the surface may gradually wash away with water, reducing their stabilizing function. When the additives are depleted, the degradation rate increases rapidly even under the same UV intensity and temperature conditions, making surface embrittlement and microcracks more likely [1–3]. In addition to the chemical factors mentioned above, physical factors such as stress acting on the material must also be considered. While stress does not directly promote chemical reactions like light, heat, or water, it can initiate and propagate macrocracks from microdefects caused by degradation. Repeated stress cycles, such as bending and vibration, can cause cracks to form at stress concentration areas*4, and damage accumulates at the tip, causing the cracks to grow, eventually leading to fracture. Therefore, stress can be said to be a factor that significantly affects the lifespan of a material [2].
3. Weather-resistance evaluation through accelerated aging testsWeatherability is the ability of a material to withstand degradation due to environmental factors such as light, heat, and water. It is an important characteristic for material selection and lifespan estimation. When assessing the weatherability of a material, natural exposure under actual operating conditions is considered to provide the most reliable information. However, this approach requires excessive time for verification, failing to match the rapid pace of material development and improvement cycles [1]. Therefore, controlled indoor aging tests of a material’s weatherability that are both accurate and rapid are essential. Current accelerated aging tests have been internationally standardized to ensure comparability of test conditions and results. A representative example is ISO (International Organization for Standardization) 4892, which specifies test methods for evaluating the weatherability of plastic materials. Test methods such as the xenon arc lamp method (ISO 4892-2) and fluorescent UV lamp method (ISO 4892-3) are specified. In Japan, a corresponding standard is JIS (Japanese Industrial Standards) K7350. These standards provide a framework for exposure while controlling environmental factors such as irradiation intensity, temperature, humidity, and wetting duration. They are widely used in Japan for material comparisons, quality control, and screening. The procedures and conditions for accelerated aging tests vary depending on the purpose of the evaluation such as material-problem identification, characteristic evaluation, or lifespan estimation. However, it is important to emphasize the reproduction in the degradation mechanism appropriate to the evaluation purpose and avoid simply increasing the stress on the material. Simply increasing the acceleration conditions may, for example, induce secondary degradation phenomena that differ from those observed in the actual environment. Therefore, it is important to set appropriate accelerated test conditions after fully considering the actual usage environment of the evaluation target and comprehensively evaluate them in combination with analyses of appearance changes, mechanical properties, and chemical indicators. We are thus currently investigating ways to improve the reproducibility of degradation phenomena that occur in the usage environment and evaluate the weathering resistance of materials in as short a time as possible. 4. Additives and analytical indicators for plastic materialsGeneral-purpose plastics, such as polypropylene, generally lack the weather resistance of their primary polymers in isolation. To address this issue, additives, such as antioxidants (primary: radical scavenging, secondary: peroxide decomposition) and light stabilizers, are blended in to improve weather resistance. Table 1 shows the types and purposes of the main additives used in industrial plastics [2]. Additives significantly affect not only the weather resistance of the plastic but also its moldability, mechanical properties, and cost. Therefore, even if the primary ingredient is the same, the properties of the material can vary greatly depending on the amount and types of additives used. To further extend the lifespan of telecommunications infrastructure, it is thus important to select the optimal material with the required weather resistance from the wide range of materials available or optimize the blend. This study focuses on weather resistance, so accelerated aging tests are essential and setting appropriate test conditions is crucial. To establish an accelerated aging test that can reproduce the degradation phenomena of real environments while accelerating the degradation process, we believe it is important to understand the degradation mechanism of materials and how acceleration conditions affect material properties. Therefore, we are currently accumulating analytical data on accelerated weathering under various conditions, using polypropylene with different additive amounts as an example.
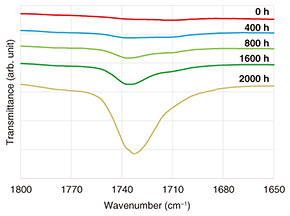
While visual observation is the most representative and provides direct information when determining the effect of test conditions on material degradation, analysis of non-visual degradation states is also important. Examples include chemical indices, such as the carbonyl index and oxidation induction time, and physical indices, such as mechanical strength (tensile strength, elongation, and modulus of elasticity), impact strength, and hardness. The carbonyl index is measured using Fourier transform infrared spectroscopy (FT-IR) and is obtained by normalizing the absorption intensity of carbonyl groups, which appears approximately around 1700–1750 cm−1, with respect to a reference peak [2]. When plastic materials are weathered by UV light or heat, carbonyl groups are generated and increase through a series of degradation reactions starting with UV absorption on the material surface. Figure 1 shows an example of FT-IR measurement results for a polypropylene sample subjected to an accelerated aging test under UV, heat, and water-spray conditions. The carbonyl group absorption peak increases with accelerated aging time. Although the carbonyl index is a relative index normalized to a reference peak, it can be measured nondestructively using FT-IR and is widely used as a useful indicator of the progress of oxidative degradation.
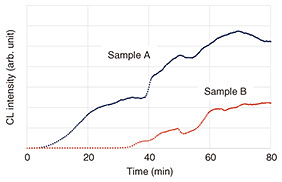
The oxidation induction time is an indicator of the time from when a sample is exposed to an oxygenated atmosphere at a constant temperature using a differential scanning calorimeter (DSC) until an oxidation reaction (heat generation) is detected [2]. In addition to DSC, oxidation induction time can also be evaluated using methods such as chemiluminescence (CL), which captures the luminescence generated during the oxidation process. Measuring the oxidation induction time allows for a relative evaluation of the effectiveness of antioxidants added to plastic materials and the resulting resistance to oxidation. Figure 2 shows typical CL measurements of the oxidation induction time for polypropylene samples with different antioxidant amounts. Sample B contains a higher amount of antioxidant than sample A, so it took longer for sample B to undergo the oxidation reaction, which indicates that the additive is effective in improving the oxidation resistance of the material. In addition to chemical indicators such as these, physical indicators, such as mechanical properties, are also important. For example, the stress-strain curve obtained from a tensile test can be used to evaluate macroscopic properties, such as elastic modulus, tensile strength, fracture strain, and toughness. There are a variety of other indicators that can be used to understand the state of degradation, and we believe that analyzing these indicators to elucidate the degradation mechanism of materials and investigate in detail their relationship with accelerated degradation conditions will lead directly to establishing optimal accelerated aging tests.
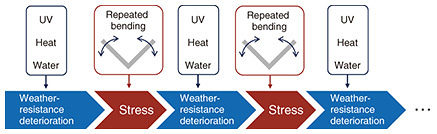
5. Accelerated aging test exampleWe introduce examples of accelerated aging testing. To evaluate the effects of stress in combination with UV, heat, and water, we conduct accelerated aging tests that simulate the periodic application of stress by repeatedly generating tensile and compressive stress through bending. Figure 3 is a conceptual diagram illustrating the cyclic conditions set for accelerated aging tests. When selecting or evaluating the benefits of plastic materials used in environments where UV, heat, water, and stress are simultaneously present, one possible method is to subject the aged samples to accelerated weathering aging under UV, heat, and water conditions then apply a mechanical load that matches the evaluation objectives and failure mode to evaluate their mechanical properties. However, in this case, because the UV, heat, water factors, and mechanical stress are applied separately, the degradation mechanism is completely different from that observed in environments where UV, heat, water, and stress are simultaneously present. Therefore, we devised an accelerated aging test that alternates between UV, heat, and water weathering blocks and stress blocks, as shown in Fig. 3. While this test method does not simultaneously apply UV irradiation, heat, water, and stress, alternating between each block is intended to gradually overlap the embrittlement and microcrack formation due to weathering with crack propagation due to stress. Figure 4 shows the appearance of a polypropylene sample subjected to an accelerated aging test under the cycle conditions shown in Fig. 3. Microcracks appear on the sample surface over time, with this being more pronounced in areas near the bends. The bending process accelerates degradation more than normal weathering. However, we will continue to optimize the weathering and bending conditions, as well as the time ratios for each block, to create an accelerated aging test that can more accurately and quickly evaluate material performance in outdoor environments where repeated stress is applied.
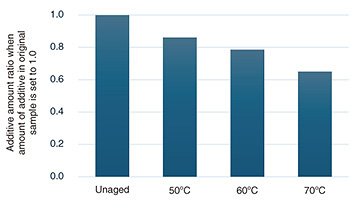
Next, we introduce an accelerated aging test that combines warm-water immersion. Warm-water immersion is carried out to accelerate the leaching of plastic additives assuming water sources such as rainfall and condensation. As mentioned above, the ISO and JIS test methods also incorporate water-induced degradation in addition to UV and heat. However, in situations where it is desired to further accelerate additive leaching, such as in environments with heavy rainfall, we believe that accelerated aging tests incorporating an immersion process may be effective. Figure 5 shows the additive reduction trend of additive-containing polypropylene when immersed in water maintained at 50–70°C for a certain period. The reduction ratio is plotted relative to the additive amount in the unaged state, which is set to 1. Comparing the amount of leaching after a certain period reveals that higher temperatures strengthen the leaching effect. By incorporating this leaching process into accelerated aging tests, for example, by immersing samples for a certain period to reduce the additive amount then weathering them, we believe it is possible to reproduce and accelerate the degradation of samples commensurate with a certain degree of additive loss. Alternatively, an accelerated aging test could be conducted by alternating weather-resistant aging blocks with immersion-induced additive leaching blocks, as in the bending test. We believe that gradually combining weather-resistant aging and water-induced additive leaching could be used to rapidly reproduce degradation in environments with heavy rainfall.
6. Future trendsWe introduced our work on accelerated aging testing to more comprehensively evaluate the weather resistance of plastic materials, with the aim of enhancing the longevity of telecommunications infrastructure. We previously established our own accelerated aging tests to evaluate the corrosion resistance and weather resistance of coatings in outdoor environments [4]. Going forward, we will continue to use this knowledge to elucidate the degradation phenomena of plastic materials and correlate accelerated aging conditions with data from real environments, with the aim of establishing accelerated-aging-test technology that is highly reproducible and rapid and can appropriately evaluate material properties. References
|
|||||||||||||||||