|
|||||||||||||
|
|
|||||||||||||
|
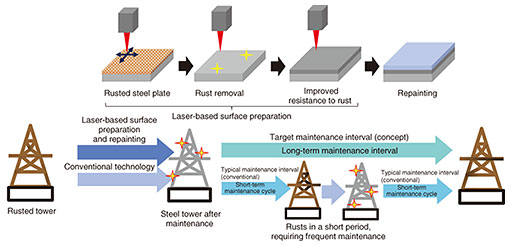
Feature Articles: Research and Development toward Sustainable Infrastructure Laser Technology for Extending the Maintenance Interval of Communication Infrastructure: Understanding the Phenomena Occurring between Rust and SteelAbstractReliable repair methods for suppressing re-deterioration are important for maintaining the long-term integrity of communication-infrastructure facilities. This article focuses on rust removal and steel-surface modification achieved through laser irradiation. It introduces our efforts to evaluate the characteristics of iron oxide, which is formed by laser irradiation, and understand its formation mechanisms, with the goal of establishing surface-preparation techniques that maintain post-repair conditions over extended periods. Keywords: extended repair interval, laser surface preparation, first-principles calculation 1. Challenges in repairing steel towers that support communication infrastructureCommunication-infrastructure facilities, including steel towers, play a vital role in ensuring the reliability and safety of communication services, which have become indispensable to modern life. NTT owns approximately 20,000 steel towers nationwide. Because these facilities are typically installed outdoors, they are exposed to wind, rain, and other environmental factors, making them susceptible to deterioration and potential loss of structural strength. To ensure long-term serviceability, regular inspection and repair are essential. Many of these facilities have been in service for decades and are aging, resulting in a yearly increase in the costs and labor required for inspections and repairs. To provide sustainable communication services, it is necessary to reduce both maintenance costs and labor demands. The current practice for maintaining steel towers, for example, involves periodic inspections to identify areas where paint has peeled or rust has progressed. When rust is found to exceed a specified threshold, maintenance personnel remove the rust and repaint the surface to suppress rust recurrence. If rust progression can be suppressed, the number of required repair operations can be reduced, helping address workforce shortage and lower costs. We aim to reduce these labor requirements and costs by improving surface-preparation methods, primarily for rust removal, so that rust recurrence and progression are suppressed and maintenance intervals are extended. Our focus is on laser-based surface preparation. Surface preparation is typically conducted using power tools and metal brushes. In contrast, laser-based methods can modify the properties of a steel surface, making it possible to create a condition that is more resistant to rust. By producing a surface to which paint adheres more strongly, long-term protection from air and water exposure can be achieved. This approach suppresses rust recurrence and progression, thus extending maintenance intervals. By reducing the number of repair operations, steel towers can be maintained at lower cost even as the available workforce declines. This article highlights laser-based surface preparation as one of the key technologies for extending maintenance cycles, as conceptually illustrated in Fig. 1.
2. Laser-based surface preparation to suppress rust and extend maintenance intervalsWe believe that the most efficient method for laser-based surface preparation that will extend maintenance intervals is to identify the substances formed on steel surfaces that are effective in suppressing recurrence and progression of rust by laser irradiation, clarify the mechanisms by which these substances are formed, and determine the optimal laser-irradiation conditions for their formation. We have adopted the following three-step approach:
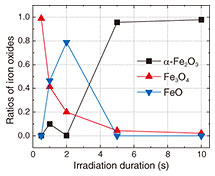
In Step 1, we irradiate rusted steel plates with a laser to remove the rust then analyze the resulting steel surface to identify the substances that have formed. In Step 2, we evaluate the work function and reaction barrier of each substance and select those with high values for both. The work function is defined as the difference between the vacuum level and Fermi level of a substance. Surfaces with higher work functions are more resistant to rust and exhibit higher corrosion potentials [1]. The reaction barrier refers to the energy required for a substance to transition from one state to another. A higher reaction barrier means that the reaction—in this case, rust formation—is less likely to occur. It follows, therefore, that selectively forming substances that exhibit both high work functions and high reaction barriers can be expected to suppress the recurrence of rust. The work function can be determined through experiments and first-principles calculations*1, while the reaction barrier can be evaluated solely through first-principles calculations. In Step 3, we use first-principles calculations to reproduce the process in which rust is removed and iron oxide is formed. From this, we identify the temperature required to form the desired substance and duration for which that temperature must be maintained. From these results, we determine the appropriate laser-irradiation duration and establish the laser technology, which is our ultimate objective. We begin by explaining each step using experimental and computational examples. Step 1: Identifying the substances formed on the steel surface after laser-based rust removalPrevious studies have shown that the rapid temperature rise caused by laser irradiation decomposes rust, and the subsequent rapid cooling leads to the formation of new iron oxides on the steel surface [2]. In this step, we identify the iron oxide species which form on the steel surface as a result of laser irradiation. To understand how laser-irradiation duration affects the proportion of the iron oxide formed, we irradiated steel with a 1.07-µm-wavelength continuous wave (CW) laser (beam diameter: 50 µm, output power: 300 W) and identified iron oxide species around the irradiated area using Raman spectroscopy*2. Figure 2 shows the proportion of iron oxides formed as a function of irradiation duration.
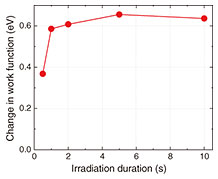
The results indicate that short irradiation durations produce a high ratio of magnetite (Fe3O4), while long durations decrease the Fe3O4 ratio. Conversely, the proportion of wüstite (FeO) increases with irradiation duration, and once the duration exceeds a certain threshold, hematite (α-Fe2O3) becomes the dominant product. This demonstrates that the iron oxide species formed on the steel surface can be controlled by adjusting the laser-irradiation duration. Step 2: Selecting substances suitable for rust suppression(1) Measuring the work function for iron oxides Figure 3 shows an example of work-function measurements for irradiated areas as a function of laser-irradiation duration. The change in work function was measured using a Kelvin probe*3. We found that the work function increases with longer irradiation durations and eventually reaches a constant value. By comparing these results with the relationship shown in Fig. 2, we infer that Fe3O4 has a relatively low work function, whereas α-Fe2O3 has a relatively high work function. Thus, increasing the proportion of α-Fe2O3 on the steel surface by extending the irradiation duration is expected to suppress rust formation.
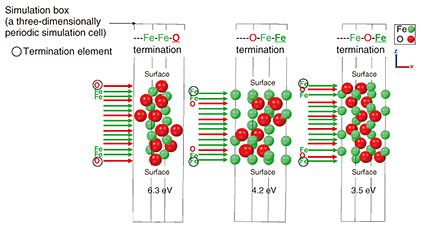
As shown in Fig. 4, α-Fe2O3 exhibits several structures with different termination, and first-principles calculations reveal that these structures yield different work functions. This suggests that controlling the crystal structure of the formed iron oxide may further enhance rust suppression. (2) Identifying the primary reaction and calculating the reaction barrier We adopt the first-principles calculation for identifying the primary reaction. There are other methods for evaluating reaction progress, e.g., observing rust progression in the air or measuring electric current during rust formation. However, rust formation on steel surfaces is believed to proceed through a chain reaction initiated by a primary reaction. These conventional methods measure the combined effects of multiple reactions, making it difficult to identify the initial reaction or compare its progression speed and reaction barrier under different conditions.
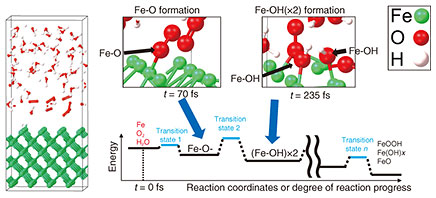
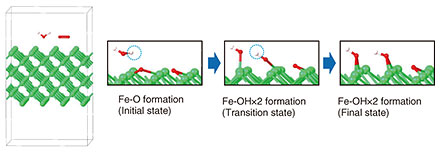
To identify the primary reaction with first-principles calculation, it is necessary to describe all possible reactions in a system composed of an Fe surface and water (H2O) and oxygen (O2) molecules. Methods capable of reproducing reactions in systems containing many molecules are suitable for this purpose. However, accurately calculating the reaction barrier for a specific reaction of interest becomes difficult in such large systems. To overcome this challenge, we devised a method with which we first investigate all reactions in a system containing many atoms and molecules, identify the primary reaction, then reproduce that reaction in a simplified model to calculate its reaction barrier. We present an example using a simplified model consisting of an Fe surface and H2O and O2 molecules. To identify the primary reaction, we first examined reactions in a system containing the Fe surface placed in a mixture of H2O and O2 molecules, as shown in Fig. 5. Using first-principles calculations, we comprehensively described all possible reactions. Figure 5 illustrates how the primary reaction of interest is identified from a multi-molecule system prior to detailed reaction-barrier calculations. We found that in the initial stage, two Fe-OH (OH: hydroxide ion) bonds form, originating from the Fe-O bond shown in Fig. 5.
We next analyzed the reaction barrier. Figure 6 shows an example in which one H2O molecule and one O2 molecule on the Fe surface are arranged to reproduce the formation of Fe-OH bonds from Fe-O bonds in a simplified system. Detailed analysis revealed that after the O2 molecule dissociates, the resulting oxygen atoms bond with Fe to form an Fe-O structure. Subsequently, the O-H (H: hydrogen) bond in H2O dissociates, and the remaining OH group adsorbs onto Fe, forming two Fe-OH bonds.
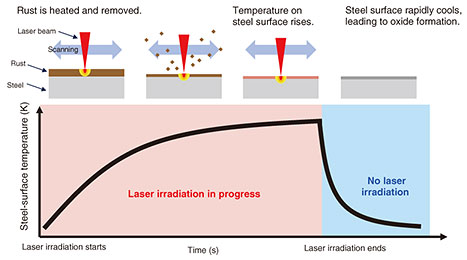
The calculated reaction barrier for the transition from Fe-O to Fe-OH was 0.40 eV, which is reasonable compared with the known reaction barriers of other substances. It has thus been difficult to calculate the dissociation and formation of multiple bonds in systems containing two or more molecules. However, by analyzing reactions in multi-molecule systems such as the one in Fig. 5, we have established a method for accurately calculating the reaction barriers of interest. Step 3: Elucidating the mechanism of substance formation and determining optimal laser-irradiation conditionsWe introduce our efforts to clarify the mechanism with which the selected substances are formed. The goal is to selectively and efficiently generate the desired substance. The rapid heating and cooling induced by laser irradiation cause iron oxide to form under non-equilibrium conditions. Figure 7 illustrates the temperature change that occurs when steel is irradiated with a laser, along with the corresponding change in the steel’s state. Laser irradiation sharply increases the temperature of the rust layer, causing it to decompose and evaporate. The steel surface then heats up and, once irradiation stops, it undergoes rapid cooling. During this non-equilibrium transition from liquid to solid, the surface reacts with O2 molecules in the air, resulting in the formation of iron oxide and other compounds.
In this process, the temperatures of the rust and steel are key controllable parameters for determining which iron oxide forms. However, temperature depends on many factors, e.g., laser power, irradiation duration, and external environment, making it time-consuming and labor-intensive to fully understand the phenomena through experiments alone. To accelerate this understanding, we use computational methods. Our approach is to use first-principles calculations to clarify microscopic processes, as described in Step 2. By combining these microscopic insights, we develop techniques to reproduce macroscopic phenomena. A critical process in Fig. 7 is what occurs after laser irradiation ends. The molten steel surface rapidly cools in air, leading to the formation of iron oxides. To reproduce the iron oxide formation that occurs during this cooling process, it is necessary to calculate the formation of periodic structures on the order of 10 nm or larger. This requires modeling phenomena that unfold over timescales exceeding 100 ps. However, first-principles calculations typically handle systems on the 1-nm scale and timescales of only ~101 picoseconds. Directly reproducing such large-scale, long-duration non-equilibrium phenomena is therefore extremely challenging. To address this, we are exploring machine-learned potential molecular dynamics [3], which use interatomic potentials trained on first-principles data. Such methods maintain accuracy close to that of first-principles calculations while dramatically reducing computational cost, enabling calculations on large spatial and temporal scales. Establishing such computational techniques will provide the foundation for controlling iron oxide formation through laser irradiation. We expect this knowledge to contribute to the development of laser technologies that suppress rust progression in various types of infrastructure, including communication facilities.
3. Summary and outlookTo ensure the sustainability of communication services, it is essential to reduce the workforce requirements and costs associated with maintaining communication infrastructure, such as steel towers. We aim to develop laser-based technology that both removes rust from steel and enhances the rust-resistance of steel, thus extending maintenance intervals and reducing associated labor and costs. Our experiments to date have shown that laser irradiation can form iron oxides that suppress rust recurrence and progression. To clarify the formation process of these oxides, we are currently developing computational techniques on the basis of first-principles calculations. Using the insights gained from this work, we will establish laser-control technologies capable of forming the desired iron oxide structures. The research effort introduced in this article represents one of the foundational technologies, approached from the perspective of laser-based surface preparation and modification, that will help extend repair cycles. Going forward, we will also consider paint durability, repainting processes, and other construction factors to develop laser technologies that contribute to extending the lifespan of communication infrastructure. References
|
|||||||||||||